Stephen B. Strum & Örn Adalsteinsson & Richard R. Black & Dmitri Segal & Nancy L. Peress & James Waldenfels
S. B. Strum
International Strategic Cancer Alliance, 538 Granite Street, Ashland, OR 97520, USA
e-mail: [email protected]
S. B. Strum
American Society of Clinical Oncology, 538 Granite Street, Ashland, OR 97520, USA
S. B. Strum
American Urological Association, 538 Granite Street, Ashland, OR 97520, USA
Ö. Adalsteinsson
International Strategic Cancer Alliance, 873 E. Baltimore Pike #333, Kennett Square, PA 19348, USA
e-mail: [email protected]
R. R. Black
Nuclear Medicine and PET Imaging, Radisphere Teleradiology Group, Beachwood, OH 44022, USA
e-mail: [email protected]
D. Segal
Valley Radiology Consultants, Poway, CA 92064, USA
e-mail: [email protected]
N. L. Peress
Life Extension Foundation, 5760 S. Scenic Drive, Sault Sainte Marie, MI 49783, USA
e-mail: [email protected]
J. Waldenfels
Life Extension Foundation, 9114 Steeplebush Court, Annandale, VA 22003-4051, USA
e-mail: [email protected]
Keywords: Dichloroacetate . DCA . non-Hodgkin’s lymphoma . NHL . PET . PET/CT . Glycolysis . Metabolomics . Warburg
Abbreviations:
DCA (dichloroacetate)
NHL (non-Hodgkin’s lymphoma)
PET (Positron Emission Tomography)
CT (computerized tomography)
FDG (fluorodeoxyglucose)
SUV (standardized uptake value)
mg (milligrams)
kg (kilograms)
R-CHOP (rituximab-Cytoxan,
Hydroxydaunomycin, Oncovin, Prednisone)
Received: 27 October 2012
Accepted: 23 November 2012
Springer Science+Business Media New York 2012
Abstract
The uptake of fluorodeoxyglucose Positron Emission Tomography in the tumors of various cancer types demonstrates the key role of glucose in the proliferation of cancer. Dichloroacetate is a 2-carbon molecule having crucial biologic activity in altering the metabolic breakdown of glucose to lactic acid. Human cell line studies show that dichloroacetate switches alter the metabolomics of the cancer cell from one of glycolysis to oxidative phosphorylation, and in doing so restore mitochondrial functions that trigger apoptosis of the cancer cell. Reports of dichloroacetate in human subjects are rare. The authors contacted individuals from Internet forums who had reported outstanding anti-cancer responses to selfmedication with dichloroacetate. With informed consent, complete medical records were requested to document response to dichloroacetate, emphasizing the context of monotherapy with dichloroacetate. Of ten patients agreeing to such an evaluation, only one met the criteria of having comprehensive clinic records as well as pathology, imaging and laboratory reports, along with single agent therapy with dichloroacetate. That individual is the focus of this report. In this case report of a man with documented relapse after state-of-the-art chemotherapy for non-Hodgkin’s lymphoma, a significant response to dichloroacetate is documented with a complete remission, which remains ongoing after 4 years. Dichloroacetate appears to be a novel therapy warranting further investigation in the treatment of cancer.
Background
The metabolic profile of malignancy has been characterized as one associated with metabolic adaptations directed to preferentially utilize pathways involved with glycolysis (Warburg et al. 1927), which in the recent literature has been termed the glycolytic phenotype of cancer. (Bui and Thompson 2006; Fang et al. 2008; Gatenby and Gawlinski 2003) In essence, this glycolytic phenotype is a Darwinian adaptation in that the cancer cell diminishes and undermines the metabolic pathways of glucose oxidation used by normal cells for energy production, and also for tumor cell elimination (Fang et al. 2008). One crucial normal cell function compromised in the battle with cancer involves mitochondrial programmed cell death or apoptosis. On the basis of the above observations, agents that target tumor metabolism, and specifically the mitochondrial ATP-producing pathways, are currently in clinical trials. Dichloroacetate (DCA) has been used over the past 30 years to treat congenital lactic acidosis—a rare metabolic disease that occurs mostly in children and young adults. (Berendzen et al. 2006; Kuroda et al. 1986; Stacpoole et al. 1997, 2008, 2006). Congenital lactic acidosis is associated with various inborn errors of mitochondrial dysfunction, and almost a thousand peer-reviewed medical publications are focused on the clinical use of DCA for this disorder. Numerous additional articles on various aspects of DCA pharmacology, metabolic effects and toxicology have also been published, but only a dozen or so papers on DCA relate to its anti-cancer activity(Bonnet et al. 2007; Bui and Thompson 2006; Cao et al. 2008; Chen et al. 2007; Christofk et al. 2008; Madhok et al. 2010; Michelakis et al. 2010, 2008; Vander Heiden 2010; Wong et al. 2008), with many of these restricted to evaluation of DCA in tumor cell lines, or in non-human animal models (Cao et al. 2008; Madhok et al. 2010; Wong et al. 2008; Sun et al. 2010). Only the publication by Michelakis et al. (2010) evaluated DCA in five human patients with glioblastoma multiforme and presented some clinical indication of response to DCA.
DCA has been reported as being an effective anti-cancer agent in vitro, and in causing regression in vivo in human glioblastoma multiforme (Michelakis et al. 2010). This, however, is the first report of a cancer patient undergoing monotherapy with DCA with the induction of a complete remission post-relapse after state-of-the-art chemotherapy with rituximab-CHOP.
Methods
Patient population
The authors SS and OA solicited all individuals who had reported favorable anti-cancer responses to DCA on Internet forums, and asked if they would agree to make their complete medical records available for detailed analysis, at no charge. Such parties were informed that the purpose of the study was to ascertain whether monotherapy with DCA could be confirmed as an active anti-cancer therapy.
Medical legal issues
Written informed consent to use detailed medical data as well as radiologic images was obtained from the subject of this case report. A copy of the written consent is available for review by the editor of this journal.
Patient exclusion criteria
Copies of all consultations and office visits, pathology reports, imaging and laboratory studies, surgical reports, radiation therapy data and chemotherapy details were requested. Of 10 possible candidates, 5 provided grossly insufficient medical records to allow for any assessment of DCA efficacy. In 3 other candidates, another treatment was commenced just before or soon after DCA was started. In 1 candidate, DCA had never been started. One of the five individuals with insufficient records was diagnosed with a non-Hodgkin’s lymphoma and appeared to have had a major response to DCA monotherapy but unfortunately he did not respond to our communications for complete medical records. In the remaining individual (TM), the subject of this report, full medical records were obtained. This individual, described in this report, was not treated by the authors.
Financial burden to participants
For every case where the initial reviews of medical records indicated a response to monotherapy with DCA, the full medical records were requested, reviewed and abstracted to an electronic health record by SS and OA. In such cases, the diagnostic pathology slides and/or tissue blocks were obtained and sent to an expert in hematopathology for a second opinion. Full imaging data in the form of Dicom files were obtained and reviewed by co-author RB, a board-certified radiologist specializing only in PET and PET/CT studies. With this approach, a highly detailed medical assessment was achieved— at absolutely no cost to any participant in this study.
Case presentation
Study subject TM is currently a 52-year old male who was first diagnosed at age 46 with non-Hodgkin’s lymphoma (NHL) on 6/1/07. He presented in late 2006 with a history of herpes zoster, low grade fevers and drenching night sweats. A 50 pound weight loss over the next 5 months occurred, and diagnoses of sinus infection and tuberculosis resulted in treatment with various drugs that included ciprofloxacin, clindamycin, INH, rifampin, pyrazimide, and other antibiotics. A 6.0 cm mass at the left lower jaw angle was noted on internal medicine evaluation on 5/1/07 in conjunction with a C-reactive protein level of 196, ESR 99, hematocrit 33.9 but a normal LDH of 138. On 5/7/07, a MRI of the neck showed multiple masses within the left parotid gland and multiple left cervical nodes up to 2.0 cm with extension into the supraclavicular region.
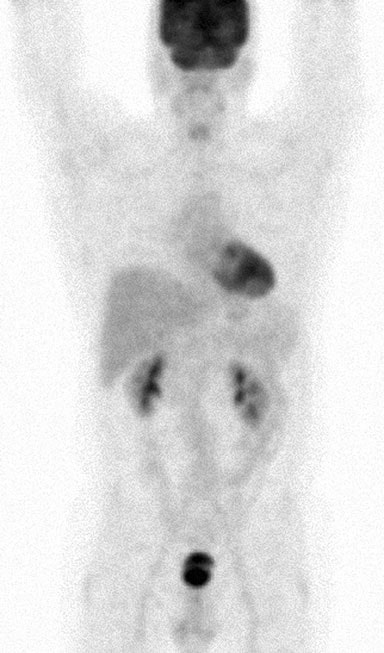
On 5/11/07, a fine needle aspiration of a 3.0 × 3.0 cm discrete mass in the left parotid gland was performed. The report and slides from this procedure were requested by the authors numerous times, but were never received. CT scans of the chest, abdomen and pelvis on 5/25/07 had revealed splenomegaly, multiple bilateral renal masses, and lymphadenopathy involving mesenteric, porta hepatis, retroperitoneal, and celiac axis nodes (Fig. 1). These findings were consistent with a diagnosis of Stage IV lymphoma (> 4 nodes involved), with anemia, and systemic symptoms of fever, night sweats, and weight loss, and with extra-nodal involvement of at least the parotid gland.
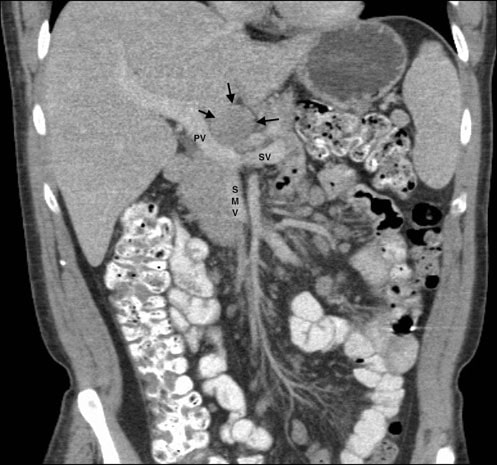
On 6/1/07, an excisional biopsy of the left parotid lesion was performed. The pathology report indicated a B-cell nonHodgkin’s lymphoma, follicular and diffuse, with focal involvement of the parotid, and classified as intermediate on the basis of cell size, morphology and proliferative index (CD71). Flow cytometry studies of the tissue samples were consistent with B-cell non-Hodgkin’s lymphoma of germinal center cell origin. Immunohistochemistry was positive for CD3, CD5, CD10, CD20, CD23, CD43, BCL-1, and BCL-2 markers. Kappa and lambda staining showed a lambda monotypic B-cell population expressing CD10 and comprising 38 % of the total tissue.
Physical examination on June 26, 2007 by a medical oncologist revealed a left parotid mass (6.0 × 5.0 cm), left jugulodigastric node (4.0 × 4.0 cm), left upper anterior cervical nodes (3.0 × 3.0 cm), right jugulodigastric node (2.0 × 2.0 cm), right and left supraclavicular nodes (3.0 × 3.0 cm), and right and left axillary nodes (2.0 × 3.0 each). Important to note was that multiple physicians examining TM reported significantly different nodal measurements of specific anatomic sites on the same day or within a matter of a few days.
A second opinion hematopathology consultation at a university medical center on 6/26/07 was suspicious for low-grade B-cell lymphoma with partial effacement of nodal architecture by an atypical and predominantly diffuse infiltration of small lymphoid cells; rebiopsy of an enlarged node was recommended. A bone marrow aspirate performed on 6/29/07 indicated a normocellular marrow with 1 large, non-paratrabecular lymphoid aggregate, containing small, mature lymphocytes. Flow cytometry on that specimen showed a clonal lambda-restricted B-cell population, characterized by small, mature cells with irregular nuclear outlines. Together, with the partial expression of CD10, and lack of CD 5 expression, a diagnosis of follicular center cell lymphoma was made along with low-grade B-cell lymphoma involving the bone marrow.
An FDG PET was done on 7/12/07 and revealed metabolic abnormalities in multiple nodal sites as well as the left parotid area. Standardized uptake value (SUV) was corrected by standardization with the liver (Wahl et al. 2009) and the findings shown in Figs. 2 and 3.
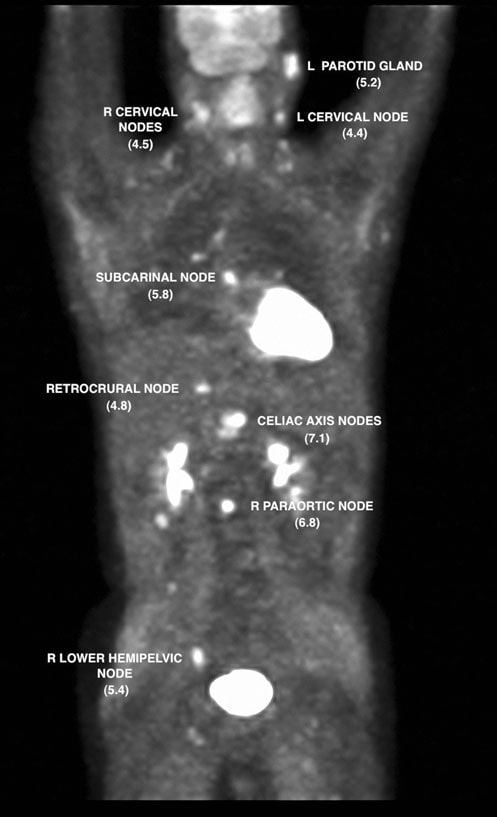
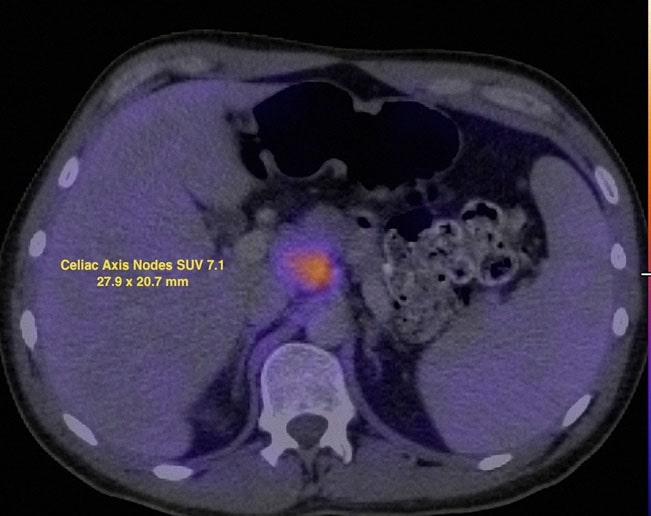
On 7/13/07, dose-dense rituximab-CHOP (R-CHOP) was started and repeated every 14 days. Cycles 2-6 were given on 7/27/07, 8/10/07, 8/24/07, 9/7/07 and 9/21/07. On 10/5/ 07, a CT of the neck, chest, abdomen and pelvis showed significant resolution of almost all previous areas of adenopathy. An FDG PET study was repeated on 10/8/07 and reported focal uptake in the R suprahilar region (Fig. 4).
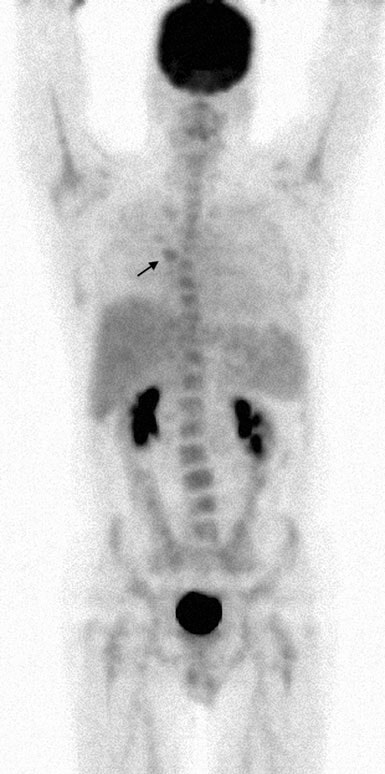
Physical examination on 10/9/07 indicated a residual fibrotic area in the left posterior cervical region measuring 4.0 by 3.0 cm. FDG PET #3 on 1/25/08 was completely normal (Fig. 5).
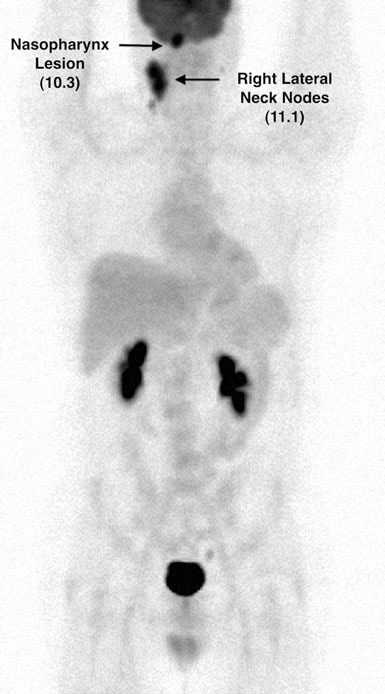
The patient was considered to be in complete remission as of 1/08. A repeat FDG PET (#4) on 4/11/08 remained normal. However, in 7/08 TM reported an 11 pound weight loss over the preceding 2 weeks, cough and sweats as well as low grade fevers. On 7/10/08 a 2.0 × 2.0 cm right anterior cervical node was found on physical examination and FDG PET #5 was obtained on 8/16/08. This showed several new hypermetabolic foci (Figs. 6 & 7).
On 9/4/08, TM was examined by a head and neck surgeon, who described a large mass of matted nodes in the right neck at levels I, 2A, 2B and upper level 5. The medical record described the patient as being extremely upset with the side effects of nausea and vomiting from the prior chemotherapy administered from 7/13/07 to 9/21/07. Per his own research he decided to embark on a treatment regimen involving DCA (dichloroacetate) and various supplements, which he began on 9/10/08. His “DCA protocol” consisted of: DCA 1,000 mg per day in one daily dose. The DCA was mixed with 10 ounces of Mountain Dew™ containing 55 mg of caffeine. TM also used vitamin B1 at 500 mg/d (through 12/10/08), alpha lipoic acid 600 mg bid, green tea (Jarrow™) 500 mg bid containing 74 mg of EGCG and 35 mg of caffeine per 500 mg.
Within 2 weeks of starting this regimen, the patient reported significant reduction in night sweats, low grade fever, anorexia and fatigue. One month after initiation of the DCA protocol, the neck nodes were noticeably smaller, and at 2 months no nodes were palpable. At 71 days into the DCA protocol, complete resolution of all systemic symptoms had occurred. TM reported a good energy level and appetite, the ability to sleep well and no side effects.
FDG PET #6 was done on 12/4/08. All abnormalities seen on the prior exam of 8/16/08 had disappeared. Via the efforts of authors SS and OA, outside expert hematopathology consultation at an international cancer center was obtained on 2/11/09. Hematoxylin and eosin stained sections showed a salivary gland to be involved by a dense atypical lymphoid infiltrate showing a vaguely nodular as well as diffuse pattern. The infiltrate was composed of small monocytoid B-lymphoid cells. Lymphoepithelial lesions were present. By morphology the differential diagnosis included extra-nodal marginal zone B-cell lymphoma (MALToma), or follicular lymphoma with monocytoid differentiation. Immunohistochemistry (IHC) studies were performed on unstained slides and were negative for BCL-2, CD3, CD43, and keratin. Positivity was seen for BCL-6 and CD10 in the vaguely nodular areas, CD20, kappa and lambda chains in some plasma cells. The final impression was reported as: “Although there is no definite IHC evidence for lymphoma, the morphology, reported flow cytometry finding of lambda monotypic B-lymphoid cells, and molecular finding of Ig Kappa gene rearrangement are consistent with a lowgrade B-cell lymphoma, which cannot be further classified.”
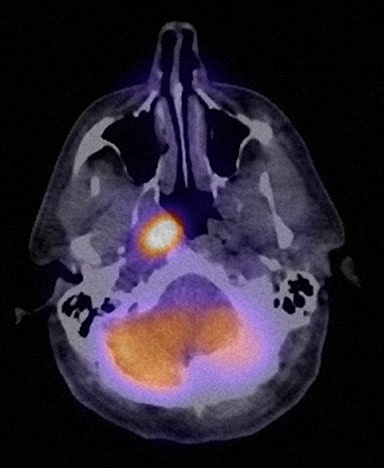
On 4/27/09, FDG PET/CT study #7 was done and showed no hypermetabolic activity. The same findings were seen on study #8 on 9/8/09. However, an internal medicine clinic note from 1/8/10 indicated that TM had noticed an enlarging neck node and physical examination confirmed a left posterior cervical node at 2 cm in diameter. In association with this he also began to have mild night sweats and fatigue. Conventional therapy was offered but declined by TM, who preferred to continue using the DCA protocol. PET/CT #9 was performed on 2/1/10 with the official report indicating right oropharyngeal uptake involving a 3.5– 4.0 cm lesion with an SUV of 5.5. Expert review by RB indicated a corrected SUV of 3.0 and a conclusion that this finding was “most consistent with normal physiologic distribution of the radiopharmaceutical.” RB also indicated that the above anatomic measurement of the oropharyngeal lesion was definitely in error. The local oncologists concluded that the patient’s lymphoma had recurred, and options for local treatment with radiation therapy as well as lenalidomide alone or with rituximab were discussed. TM again refused further chemotherapy and continued on DCA. Hematology-oncology follow-up on 1/25/11 indicated a 2.0 cm left cervical lymph node. Again, options for further treatment were discussed and included bendamustine, fludarabine, radioimmunotherapy with Zevalin but all were refused. On 4/10/11, FDG PET/CT #10 was performed and showed no evidence of hypermetabolic activity (Fig. 8).
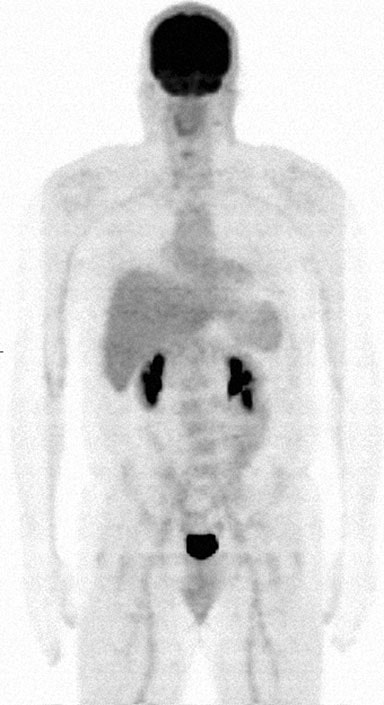
As of November 2012, TM reports that he is fine and without systemic symptoms. He notes waxing and waning of small nodes in his neck which he says are pea-sized. He reports continued use of his DCA protocol but states that he decreased the frequency of DCA dosing to three times per week. He states that his energy level is excellent, that he is working full time, but because he cannot get insurance he has not had further medical evaluation by physical examination, laboratory testing or any imaging studies. The authors SS and OA have facilitated recent FDG PET/CT imaging on 11/8/12 and laboratory testing on 11/ 9/12, at no charge to TM. The FDG study was completely normal. All laboratory tests, other than a lipid elevation of triglycerides and LDL, are normal, including biomarkers such as β-2 μglobulin, soluble IL-2 receptors, and ESR.
Discussion
Tumor cells have a remarkably different metabolism compared to the normal cells from which they are derived. They exhibit an altered metabolism that allows them to sustain higher proliferative rates and resist various apoptotic signals (Bonnet et al. 2007; Bui and Thompson 2006). This altered and increased metabolism means cancer cells require large quantities of protein, lipid and nucleotides, as well as energy in the form of ATP. Almost 100 years ago, Otto Warburg published his findings showing that cancer cells demonstrate an alteration in cellular energy involving enhanced aerobic glycolysis. (Warburg 1956a, b; Warburg et al. 1927)
One of the proposed mechanisms of action of DCA is to shift tumor cell metabolism from the glycolytic cytoplasmbased pathway, preferred by tumor cells, to a mitochondrialbased glucose oxidative (oxidative phosphorylation) pathway (Bonnet et al. 2007). In doing so, DCA restores the mitochondria’s apoptotic function and reverses the metabolic alterations that have allowed tumor cells to survive, proliferate, and metastasize. This posited action of DCA is associated with its ability to reverse the inhibition of PDH (pyruvate dehydrogenase)—a crucial enzyme that allows pyruvate entry into the Krebs Cycle via its oxidation to the key substrate acetyl CoA. DCA does this by inhibiting mitochondrial PDK (pyruvate dehydrogenase kinase). Therefore, DCA inhibits the inhibitor involved with PDH production. DCA accomplishes this by preventing the phosphorylation and inactivation of PDH by PDK. (Constantin-Teodosiu et al. 1999)
Cancer cell aggressiveness has been associated with the degree of hyperpolarization of the mitochondrial membrane potential (Heerdt et al. 2005). Bonnet et al. have defined an array of mitochondrial effects of DCA, as follows. DCA has a rapid and dose-dependent effect leading to mitochondrial membrane depolarization that occurs within 5 min. Due to this aspect of DCA’s activity, AIF (apoptosis inducing factor) is translocated to the nucleus of the cell and cytochrome C becomes diffusely present in the cytoplasm. DCA dose-dependently increases H2O2 production within Complex I of the ETC. H2O2 generation activates Kv1.5 channels which in turn inhibits NFAT1, a known inhibitor of apoptosis. Other biochemical effects of DCA include increasing the expression of annexin, activating caspases 3 & 9, decreasing survivin and PCNA (proliferating cell nuclear antigen) (Bonnet et al. 2007). Additional studies have shown that upregulation of Kv1.5 channels likely decreases cellular potassium, which activates caspases and apoptosis (Remillard and Yuan 2004; Wang 2004; Pan and Mak 2007).
There have been few studies of the effect of DCA on human cancer growth, and most of these have been in vitro. Tong et al. studied DCA alone or in combination with 5-FU against four human colorectal cancer cell lines. They showed DCA to be synergistic with 5-FU in inducing apoptosis, inhibiting cancer cell proliferation, enhancing cell cycle arrest at G1/S phase, decreasing Bcl-2 expression, while increasing the expression of Bax and caspase-3 (Tong et al. 2011). The study of DCA by Michelakis et al. (2010) in five patients with glioblastoma multiforme describes radiologic regression and provides images of two patients with an apparent response to DCA, although debulking of tumor in patient number 2 seems to confound the interpretation of the DCA effect.
F-18 Fluorodeoxyglucose (F-18 FDG) PET is now being used by an increasing number of oncologists to determine the extent of malignancy and to document the therapeutic response and provide prognostic information after anti-neoplastic therapy. The patient presented had a baseline FDG PET consistent with his clinical signs and symptoms of a highly active neoplastic process. After treatment with state of the art chemotherapy using R-CHOP, he achieved a complete remission of short duration; his relapse was again characterized by abnormal FDG PET abnormalities. However, his response to DCA monotherapy led to a durable complete remission, now at 4 years. The uptake of F18 FDG by neoplasms is regarded as an example of the Warburg effect i.e., the utilization of glucose by tumors in the presence of oxygen (aerobic glycolysis). However, the singular functionality of FDG PET as seen in various cancers results from multiple factors, some of which are:
- Phosphorylation of F18 FDG to F18 FDG-6 phosphate by Hexokinase (HK)
- Up-regulation of HK by hypoxia-inducible transcription factors e.g., HIF-1α (Mathupala et al. 1997, 2001)
- Over-expression of HK, especially the isomer HKII, in many cancer cell populations
- Crucial role of HK to tumor cell bioenergetics i.e., Warburg effect (Bustamante et al. 1981; Bustamante and Pedersen 1977)
- Absence of glucose-6-phosphatase (G6Pase) in highly malignant, rapidly growing tumors
- Resultant trapping of F18-FDG-6 phosphate by cancer cells (Higashi et al. 2002)
In the study subject we have presented, a complete remission was documented by FDG PET on 12/4/08 and has continued 4 years without any therapeutic intervention other than continued use of DCA, but at a lower dosing frequency of three times per week. As of 12/5/12, TM reports no significant findings of peripheral neuropathy at the dose of ≤ 10 mg/kg/day, which is consistent with the findings of Michelakis et al. (2010) who reported no significant peripheral neuropathy with DCA doses of less than 6.25 mg/kg twice a day. TM also has had no other side effects.
An explanation posited for the dramatic response documented in this case report is that of spontaneous remission of a low to intermediate grade NHL. This seems improbable in light of the dramatic reduction and disappearance of systemic symptoms and adenopathy within weeks of starting DCA. In addition, the relatively quick relapse after initial chemotherapy with R-CHOP makes spontaneous remission an unlikely occurrence in TM’s illness.
Our study, presented herein, should provoke in-depth discussion about the use of metabolic therapy such as DCA in cancer patients. Other issues which seem pertinent to this report include the possible role of a low glycemic diet in the prevention and treatment of cancer, and the need for focusing on the methodology and reporting format involved with PET imaging. The utilization of medical records from individuals exploring anticancer therapy without the oversight of a healthcare practitioner is a unique approach in medical detective work. Obtaining patient medical records, as well as DVDs and reports of imaging, pathology reports and slides, and laboratory reports is challenging and time consuming. However, if further study of human subjects confirms that DCA has significant anti-cancer activity, the approach used in this investigation should be considered on a larger scale.
Conclusion
The outstanding response to DCA monotherapy of patient TM, in a context of relapse after R-CHOP therapy, warrants further clinical study of DCA in human patients with various malignancies, especially given the negligible cost and minimal toxicity of this simple 2-carbon molecule.
Acknowledgments
The authors wish to thank Life Extension Foundation in Fort Lauderdale, Florida for financial assistance, and also Robert Vergara of Life Extension for help optimizing graphic files used in this report. Jim Tassano was instrumental in alerting authors SS and OA about reports of significant clinical responses in cancer patients receiving DCA.
Competing Interests
The authors declare they have no financial or non-financial competing interests.
Authors’ Contributions
SS, OA, NP, JW contributed in the writing of the manuscript. SS and OA obtained all medical records, facilitated pathology review, compiled electronic health records, and followed up with study participants. RB reviewed all PET and PET/CT images, provided corrected SUVs and measurements. DS facilitated obtaining key DVDs of imaging studies and rendering radiologic opinion.
Author’s Information
SS is a board-certified medical oncologist with over 40 years of clinical practice as well as author of many peer-reviewed articles on Hodgkin’s disease, prostate cancer and supportive care of the cancer patient.
REFERENCES
1 Berendzen K, Theriaque DW, Shuster J, Stacpoole PW (2006) Therapeutic potential of dichloroacetate for pyruvate dehydrogenase complex deficiency. Mitochondrion 6:126–135
2Bonnet S, Archer SL, Allalunis-Turner J, Haromy A, Beaulieu C, Thompson R et al (2007) A mitochondria-K+ channel axis is suppressed in cancer and its normalization promotes apoptosis and inhibits cancer growth. Cancer Cell 11:37–51
3Bui T, Thompson CB (2006) Cancer’s sweet tooth. Cancer Cell 9:419– 420
4Bustamante E, Pedersen PL (1977) High aerobic glycolysis of rat hepatoma cells in culture: role of mitochondrial hexokinase. Proc Natl Acad Sci U S A 74:3735–3739
5Bustamante E, Morris HP, Pedersen PL (1981) Energy metabolism of tumor cells. Requirement for a form of hexokinase with a propensity for mitochondrial binding. J Biol Chem 256:8699–8704
6Cao W, Yacoub S, Shiverick KT, Namiki K, Sakai Y, Porvasnik S et al (2008) Dichloroacetate (DCA) sensitizes both wild-type and over expressing Bcl-2 prostate cancer cells in vitro to radiation. Prostate 68:1223–1231
7Chen Z, Lu W, Garcia-Prieto C, Huang P (2007) The Warburg effect and its cancer therapeutic implications. J Bioenerg Biomembr 39:267–274
8Christofk HR, Vander Heiden MG, Harris MH, Ramanathan A, Gerszten RE, Wei R et al (2008) The M2 splice isoform of pyruvate kinase is important for cancer metabolism and tumour growth. Nature 452:230–233
9Constantin-Teodosiu D, Simpson EJ, Greenhaff PL (1999) The importance of pyruvate availability to PDC activation and anaplerosis in human skeletal muscle. Am J Physiol 276:E472–E478
10Fang JS, Gillies RD, Gatenby RA (2008) Adaptation to hypoxia and acidosis in carcinogenesis and tumor progression. Semin Cancer Biol 18:330–337
11Gatenby RA, Gawlinski ET (2003) The glycolytic phenotype in carcinogenesis and tumor invasion: insights through mathematical models. Cancer Res 63:3847–3854
12Heerdt BG, Houston MA, Augenlicht LH (2005) The intrinsic mitochondrial membrane potential of colonic carcinoma cells is linked to the probability of tumor progression. Cancer Res 65:9861–9867
13Higashi T, Saga T, Nakamoto Y, Ishimori T, Mamede MH, Wada M et al (2002) Relationship between retention index in dualphase (18)F-FDG PET, and hexokinase-II and glucose transporter-1 expression in pancreatic cancer. J Nucl Med 43:173–180
14Kuroda Y, Ito M, Toshima K, Takeda E, Naito E, Hwang TJ et al (1986) Treatment of chronic congenital lactic acidosis by oral administration of dichloroacetate. J Inherit Metab Dis 9:244–252
15Madhok BM, Yeluri S, Perry SL, Hughes TA, Jayne DG (2010) Dichloroacetate induces apoptosis and cell-cycle arrest in colorectal cancer cells. Br J Cancer 102:1746–1752
16Mathupala SP, Rempel A, Pedersen PL (1997) Aberrant glycolytic metabolism of cancer cells: a remarkable coordination of genetic, transcriptional, post-translational, and mutational events that lead to a critical role for type II hexokinase. J Bioenerg Biomembr 29:339–343
17Mathupala SP, Rempel A, Pedersen PL (2001) Glucose catabolism in cancer cells: identification and characterization of a marked activation response of the type II hexokinase gene to hypoxic conditions. J Biol Chem 276:43407–43412
18Michelakis ED, Webster L, Mackey JR (2008) Dichloroacetate (DCA) as a potential metabolic-targeting therapy for cancer. Br J Cancer 99:989–994
19Michelakis ED, Sutendra G, Dromparis P, Webster L, Haromy A, Niven E et al (2010) Metabolic modulation of glioblastoma with dichloroacetate. Sci Transl Med 2:31ra34
20Pan JG, Mak TW (2007) Metabolic targeting as an anticancer strategy: dawn of a new era? Sci STKE 2007:14
21Remillard CV, Yuan JX (2004) Activation of K+ channels: an essential pathway in programmed cell death. Am J Physiol Lung Cell Mol Physiol 286:L49–L67
22Stacpoole PW, Barnes CL, Hurbanis MD, Cannon SL, Kerr DS (1997) Treatment of congenital lactic acidosis with dichloroacetate. Arch Dis Child 77:535–541
23Stacpoole PW, Kerr DS, Barnes C, Bunch ST, Carney PR, Fennell EM et al (2006) Controlled clinical trial of dichloroacetate for treatment of congenital lactic acidosis in children. Pediatrics 117:1519–1531
24Stacpoole PW, Gilbert LR, Neiberger RE, Carney PR, Valenstein E, Theriaque DW et al (2008) Evaluation of long-term treatment of children with congenital lactic acidosis with dichloroacetate. Pediatrics 121:e1223–e1228
25Sun RC, Fadia M, Dahlstrom JE, Parish CR, Board PG, Blackburn AC (2010) Reversal of the glycolytic phenotype by dichloroacetate inhibits metastatic breast cancer cell growth in vitro and in vivo. Breast Cancer Res Treat 120:253–260
26Tong J, Xie G, He J, Li J, Pan F, Liang H (2011) Synergistic antitumor effect of dichloroacetate in combination with 5-fluorouracil in colorectal cancer. J Biomed Biotechnol 2011:740564
27Vander Heiden MG (2010) Targeting cell metabolism in cancer patients. Sci Transl Med 2:31ed31
28Wahl RL, Jacene H, Kasamon Y, Lodge MA (2009) From RECIST to PERCIST: evolving considerations for PET response criteria in solid tumors. J Nucl Med 50(Suppl 1):122S–150S
29Wang Z (2004) Roles of K+ channels in regulating tumour cell proliferation and apoptosis. Pflugers Arch 448:274–286
30Warburg O (1956a) On respiratory impairment in cancer cells. Science 124:269–270 Warburg O (1956b) On the origin of cancer cells. Science 123:309–314
31Warburg O, Wind F, Negelein E (1927) The metabolism of tumors in the body. J Gen Physiol 8:519–530
32Wong JY, Huggins GS, Debidda M, Munshi NC, De Vivo I (2008) Dichloroacetate induces apoptosis in endometrial cancer cells. Gynecol Oncol 109:394–402
Related content: